
One-third of Rapid Reviews in Ireland involve price negotiations to avoid a HTA
Rapid Reviews in Ireland Systematic reviews have long been a key component of evidence-based medicine. The use of methods to expedite systematic reviews is ever-increasing due to time and resource constraints. Systematic reviews typically take at lea...
READ MORE
Health Technology Assessment: An In-Depth Analysis
Health Technology Assessments (HTA’s) Health technology assessment (HTA) is a multidisciplinary process that evaluates the clinical effectiveness, safety, economic viability, and broader impact of health technologies. Health technologies refer to a...
READ MORE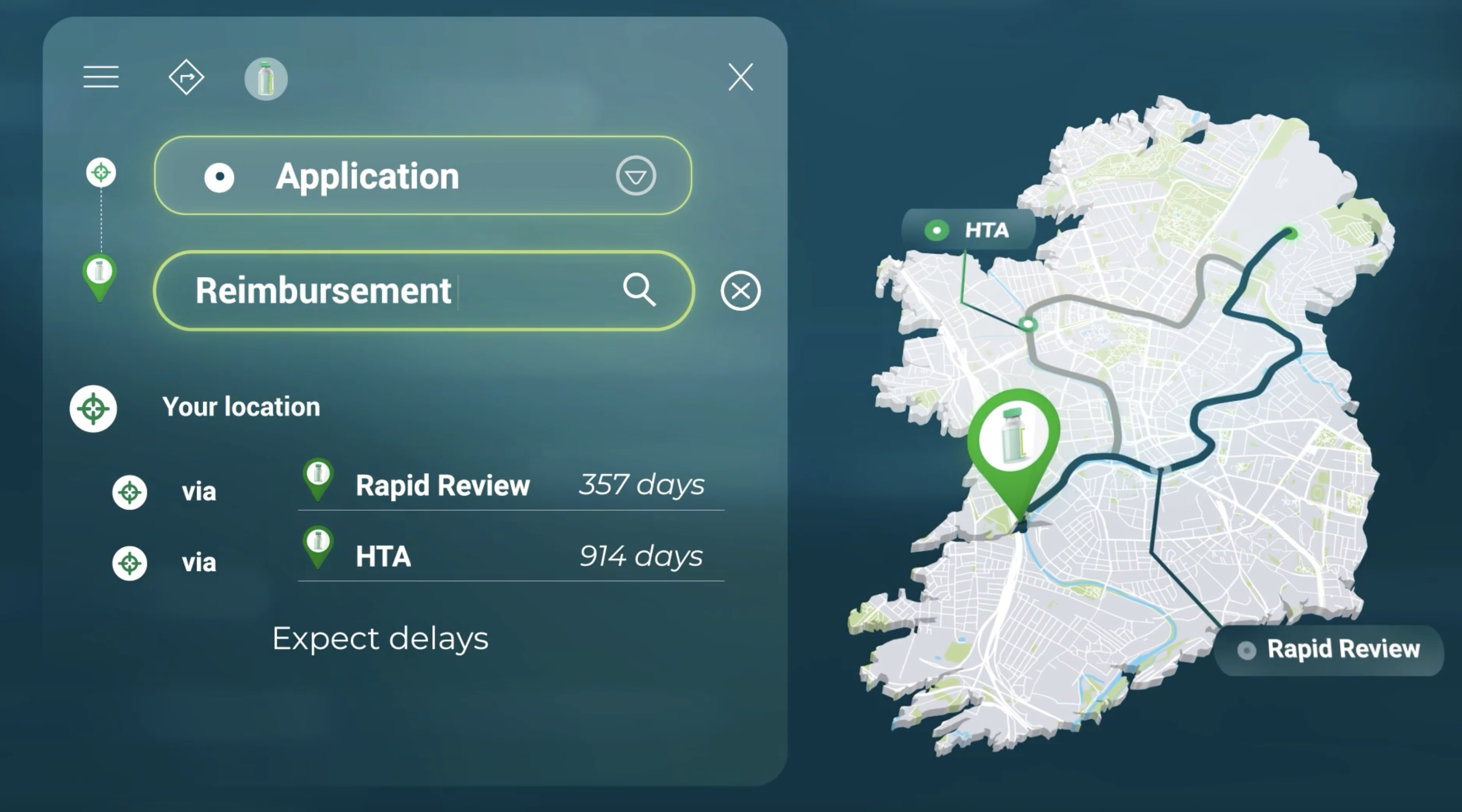
Understanding the Irish Reimbursement Process for Medicines
If you’re a pharmaceutical company looking to introduce a new medicine to the Irish market, navigating the reimbursement process can be complex and time-consuming. In this video you’ll gain a better understanding of the two routes to reimbursem...
READ MORE
Unlocking Market Potential: How Market Access Consultancy Services Can Accelerate Your Medicine Product Launch
In the rapidly evolving healthcare industry, market access is crucial for the successful launch of new products and services. Market access consultancy services play a pivotal role in helping organisations navigate the complexities of market entry, p...
READ MORE
We are attending ISPOR in Vienna
This must-attend event welcomes all healthcare stakeholders and is directly relevant to researchers and academicians, assessors and regulators, payers and policymakers, the life sciences industry, healthcare providers, and patient engagement organiza...
READ MORE